DATE2025.07.19 #Press Releases
Redesigning Natural Product Scaffolds Opens New Avenues in Medium-Sized Macrocyclic Drug Discovery
— Strategic Reengineering of the Anticancer Agent Ecteinascidin 743 Leads to Potent Macrocyclic Derivatives —
Summary
Assistant Professor Ryo Tanifuji, Erina Hosono (at the time of research: Master's student), and Hisae Kamakura (at the time of research: technical assistant) from the Graduate School of Science at The University of Tokyo, along with Professor Hiroki Oguri, collaborated with graduate student Satoshi Yoshida and Project Professor Sota Sato from the Graduate School of Engineering at The University of Tokyo, as well as with Chief Research Assistant Yukiko Muramatsu and Director Hiroyuki Seimiya from the Division of Molecular Therapeutics at the Cancer Chemotherapy Center of the Japanese Foundation for Cancer Research, and Research Assistant Yoshimi Ohashi and Director Shingo Dan from the Division of Molecular Pharmacology at the same center.
The team developed a synthetic strategy for scaffold redesign of ecteinascidin 743 (also known as trabectedin), a marine-derived anticancer natural product.
This approach enabled the efficient generation of structurally diverse macrocyclic medium-sized molecules, combining high molecular complexity with potent anticancer activity.
The resulting derivatives retained DNA double-strand cleavage activity and demonstrated sub-nanomolar growth inhibition across a panel of 39 human cancer cell lines (JFCR39).
Notably, variations in macrocyclic architecture markedly altered activity profiles across cancer types.
These findings offer a new molecular design framework for the development of anticancer agents derived from complex natural products.
This approach enabled the systematic reengineering of the macrocyclic segment by repositioning the bridging position on the core scaffold (from C1–C4 to C1–C5), allowing access to previously unexplored skeletal architectures.
Starting from the natural product cyanosafracin B, they synthesized a library of more than ten macrocyclic medium-sized molecules (14- to 17-membered rings) in just 6–10 steps—approximately one-third the length of traditional semi-synthetic routes of ecteinascidin 743.
The resulting compounds retain the essential pharmacophore responsible for DNA alkylation and induce DNA double-strand breaks in cancer cells.
Several derivatives demonstrated sub-nanomolar growth inhibition across a panel of 39 human cancer cell lines (JFCR39).
Differences in ring size and functional group incorporation modulated biological response profiles, including shifts in mechanisms of action and cell-line selectivity.
These findings highlight the potential of scaffold-level redesign to expand conformational and pharmacological space, offering a promising strategy for medium-sized drug discovery inspired by complex natural products.
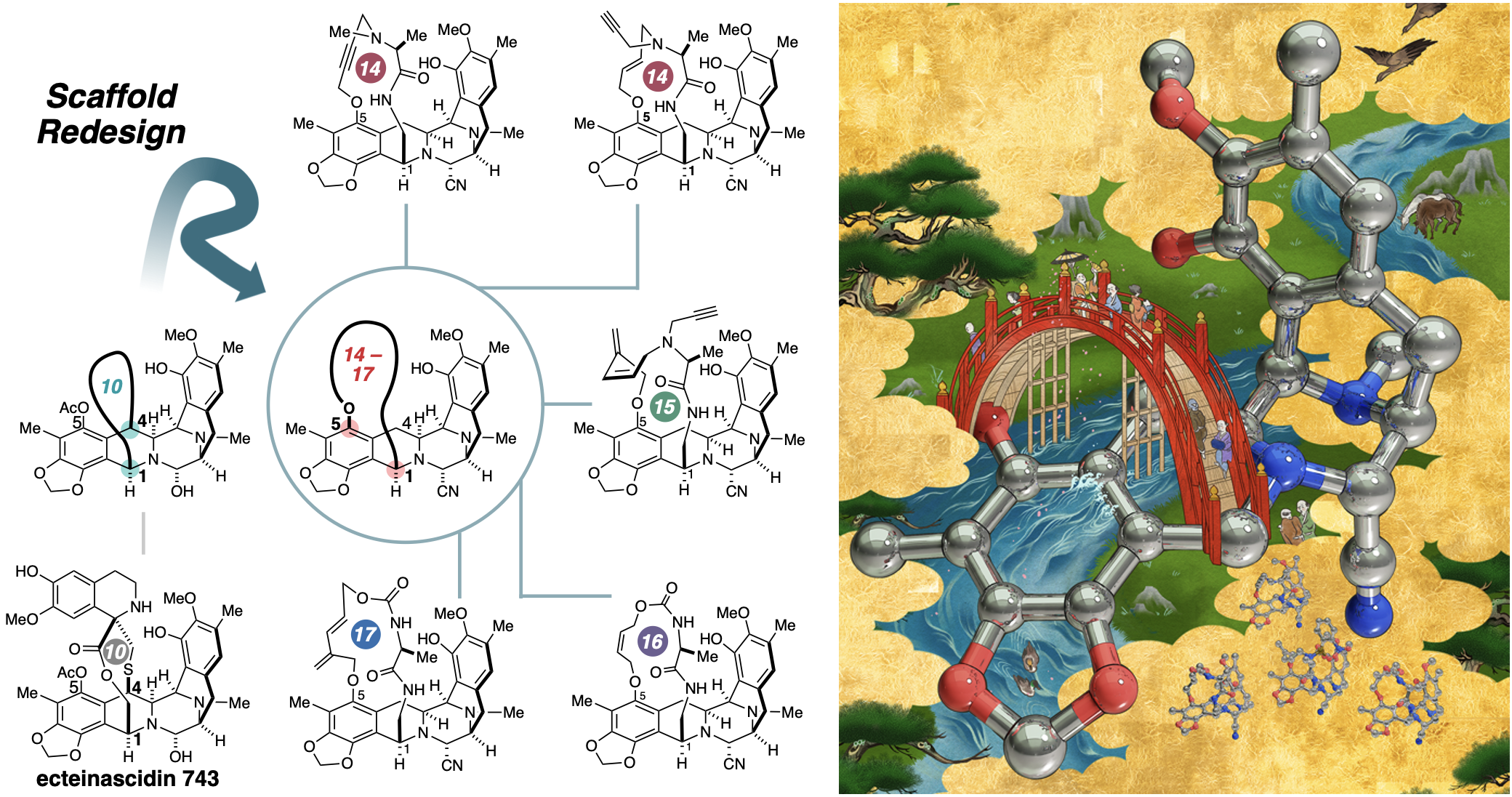 Figure: Development of Antitumor Macrocyclic Medium-Sized Molecules through Strategic Redesign of Natural Product Scaffolds
Figure: Development of Antitumor Macrocyclic Medium-Sized Molecules through Strategic Redesign of Natural Product Scaffolds
Links
The Japanese Foundation for Cancer Research (JFCR)
Journals
-
Journal name ChemTitle of paper Strategic Scaffold Redesign of Ecteinascidins: An Approach for Generating Anticancer Macrocycles


