DATE2025.09.16 #Press Releases
Revolutionizing Chemical Synthesis in Water
-- Carbon Nanotubes Step Up as Catalysts’ “Smart Sidekick” --
Presentation points
- A long-standing challenge has been overcome: asymmetric synthesis in water using solid catalysts is now a reality. This breakthrough paves the way for greener production of pharmaceuticals and high-performance chemicals.
- The team utilized single-walled carbon nanotubes (SWNTs)—a material previously underutilized in this context—to immobilize catalysts. SWNTs act not just as passive supports but as “active enablers,” stabilizing and allowing reuse of the catalysts.
- The new method avoids covalent bonding, enabling simpler and more efficient catalyst design. It marks a significant stride toward environmentally friendly chemical synthesis.

Catalyst Design Overview
Overview
A research team led by Assistant Professor Taku Kitanosono and Project Professor Shū Kobayashi at the Graduate School of Science, The University of Tokyo, has developed a novel technique for catalytic asymmetric synthesis ( Note 1)in water —a long-standing challenge in synthetic chemistry.
The method employs single-walled carbon nanotubes (SWNTs)( Note 2 ), ultra-thin cylinders of carbon successfully mass-produced by Japanese industry. By adsorbing specially designed chiral Lewis acid catalysts( Note 3)onto the surface of SWNTs, the team achieved stable immobilization without relying on covalent bonding.
Conducting chemical reactions in water is a cornerstone of green chemistry, offering safer and more sustainable alternatives to traditional methods. However, conventional chiral Lewis acid catalysts are notoriously water-sensitive, often losing activity rapidly in aqueous environments.
Previous approaches focused on covalent immobilization to prevent catalyst leaching, but this came at the cost of design complexity and reduced flexibility. In contrast, the new technique leverages the electronic and surface properties of SWNTs, enabling noncovalent immobilization that maintains catalytic activity even after ten reuse cycles.
This breakthrough opens new avenues for the eco-friendly production of pharmaceuticals and advanced chemical materials, demonstrating that SWNTs are not just passive supports but active partners in catalysis.
Announcements
Background
Using water as a reaction medium in chemical synthesis is a key strategy for reducing environmental impact and promoting a more sustainable society. In fields such as pharmaceuticals and high-performance chemical manufacturing, this principle—known as green chemistry—is increasingly in demand.
However, performing reactions in water presents significant technical hurdles. In particular, chiral Lewis acid catalysts tend to deactivate upon contact with water or other reactive species, leading to poor reaction outcomes and diminished stereoselectivity (Figure 1A).
To address this, previous studies have relied on covalent immobilization of catalysts, which prevents leaching and maintains performance. Yet this approach comes with a trade-off: the process of designing and synthesizing such catalysts becomes more complex, limiting flexibility and scalability (Figure 1B).
In this context, the present study introduces a noncovalent immobilization strategy using single-walled carbon nanotubes (SWNTs), achieving stable catalyst performance without the need for covalent bonding (Figure 1C). This marks a paradigm shift in how catalysts can be supported and reused in aqueous environments.

Figure 1: (A) Mechanism of deactivation and loss of stereoselectivity of chiral Lewis acid catalysts in water
(B) Conventional immobilization methods for chiral metal catalysts
(C) First demonstration of noncovalent immobilization of chiral Lewis acids on SWNTs in this study
Research Details
In this study, the team set out to develop a method for immobilizing catalysts without relying on covalent bonding—a bold challenge in the field of synthetic chemistry.
The key to their success was the use of single-walled carbon nanotubes (SWNTs). These ultra-thin, tubular carbon structures possess exceptional electrical and structural properties. The researchers hypothesized that the surface of SWNTs could help stabilize catalysts in aqueous environments.
Through experimentation, they demonstrated that simply adsorbing a custom-designed scandium complex onto the surface of SWNTs was sufficient to create a functional immobilized catalyst (Figure 1C). This process harnesses π–π interactions—a type of attractive force between the catalyst and the nanotube surface. These interactions helped fine-tune the electronic environment of the catalyst, resulting in enhanced stability and performance.
The catalyst system showed high activity in key reactions conducted in water. Remarkably, even after ten consecutive uses, the catalyst retained its performance, with minimal leaching or degradation—comparable to systems immobilized via covalent bonding.
When compared with other carbon-based materials such as carbon black (CB) and multi-walled carbon nanotubes (MWNTs), the superiority of SWNTs became clear (Figure 2). In those cases, catalytic activity dropped quickly or failed to initiate reactions altogether.
These findings reveal that SWNTs are far more than passive supports—they act as active partners, enhancing the function of the catalyst itself. Moreover, by chemically modifying the SWNT surface or adjusting the molecular structure of the catalyst, the team found they could further improve performance. This opens up exciting possibilities for future catalyst design.
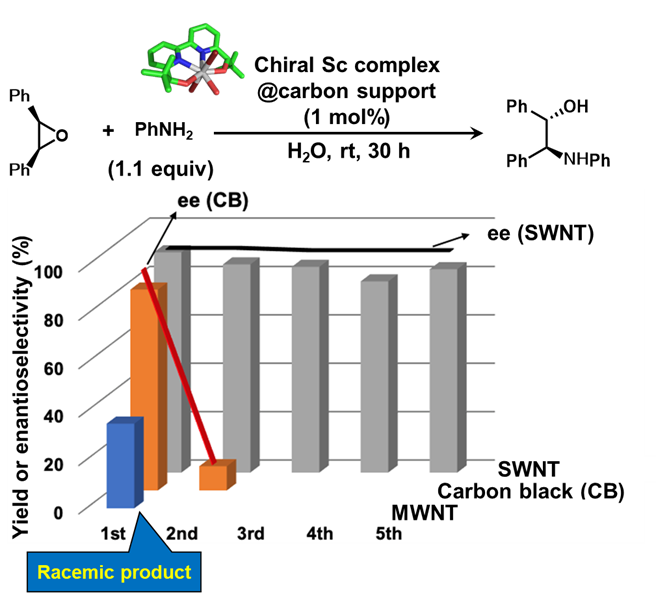
Figure 2: Preliminary results demonstrating the effect of different carbon supports on catalytic performance
Future Outlook
This study represents a major breakthrough in overcoming the long-standing challenge of conducting chemical reactions in water—a feat achieved through a novel and elegant approach.
By harnessing the unique properties of single-walled carbon nanotubes (SWNTs), the researchers succeeded in stabilizing high-performance catalysts without relying on covalent bonding. This opens the door to designing catalysts that are not only more efficient but also easier to tailor for specific applications.
Looking ahead, this technology holds great promise for transforming the manufacturing processes of pharmaceuticals and fine chemicals, making them more environmentally friendly and cost-effective.
Ultimately, this research demonstrates how the synergy between materials science and synthetic chemistry can lead to innovative solutions that contribute to a more sustainable future.
Related Information
Researcher and Contact Information
The University of Tokyo
Graduate School of Science, Department of Chemistry
Synthesis Organic Chemistry Laboratory
Taku Kitanosono, Assistant Professor
Presidential Endowed Chair for “Green Material Conversion
Shū Kobayashi, Project Professor
(also Department of Chemistry, Graduate School of Science)
Publications
-
Journal name Chemical Science Title of paper Noncovalent Immobilization of Chiral Lewis Acids on Single-Walled Carbon Nanotubes as a Tool for Synthetic Organic AquachemistryAuthor(s) Taku Kitanosono,* Satoshi Tanaka, Dongxin Zhang, Tomoya Hisada, Yasuhiro Yamashita and Shū Kobayashi* (*Corresponding authors) DOI Number 10.1039/D5SC05390K
Funding Acknowledgment
This research was supported by the following grants:
Grant-in-Aid for Scientific Research on Innovative Areas
“Development of Ubiquitin-Selective Recognition and Labeling Methods Using Polymers”
Project Number: JP19H05288
Grant-in-Aid for Young Scientists
“Noncovalent Immobilization Strategy for Chiral Lewis Acid Catalysts Using π-Electronic Materials”
Project Number: JP20K15272
Grant-in-Aid for Scientific Research (C)
“Catalyst Development Utilizing the Electronic Properties of Single-Walled Carbon Nanotubes”
Project Number: JP25K08633
Grant-in-Aid for Scientific Research (S)
“Establishing Organic Chemistry Centered on Water: Aquachemistry”
Project Number: JP22H04972
Glossary
Note 1 Catalytic Asymmetric Synthesis
A method for synthesizing enantiomerically enriched compounds using only a small amount of a chiral source. This technique allows for the theoretical production of an unlimited number of enantiomers. It was recognized with the 2001 Nobel Prize in Chemistry awarded to Ryōji Noyori and two other scientists. Enantiomers are molecules that are non-superimposable mirror images of each other—like left and right hands. The term chirality refers to this property. ↑up
Note 2 Single-Walled Carbon Nanotubes (SWNTs)
Discovered in 1991 by Dr. Sumio Iijima, carbon nanotubes are categorized into multi-walled and single-walled types based on their structure. SWNTs are known for their unique properties—lightweight, high strength, flexibility, excellent electrical and thermal conductivity, and semiconducting behavior—that far surpass those of conventional materials. Although mass production was once a bottleneck, ZEON Corporation began operating a commercial-scale SWNT production facility in 2015, spurring global efforts toward industrialization.↑up
Note 3 Chiral Lewis Acid Catalysts
Catalysts that enhance the reactivity of electron-rich compounds such as carbonyls by accepting lone pairs of electrons. These are typically cationic metal complexes. When chiral ligands are used in the metal complex, the resulting catalyst can control the stereochemical environment, enabling asymmetric reactions. This approach—using a small amount of chiral source to convert achiral molecules into optically active ones—is a cornerstone of modern organic synthesis. ↑up


