DATE2025.06.26 #Press Releases
Elucidation of the G Protein Selectivity Mechanism of the Parathyroid Hormone Type 1 Receptor
— Providing a Foundation for the Development of Next-Generation Osteoporosis Treatments —
Summary
A research team led by Project Assistant Professor Fumiya Sano and Professor Osamu Nureki at the School of Science, The University of Tokyo; graduate student Kota Shimizume, Associate Professor Masataka Yanagawa, and Professor Asuka Inoue at the Graduate School of Pharmaceutical Sciences, Kyoto University; and Project Researcher Kazuhiro Kobayashi at the Research Center for Advanced Science and Technology, The University of Tokyo, has elucidated the molecular mechanism by which the parathyroid hormone type 1 receptor (PTH1R) selectively engages with specific G proteins from among multiple candidates.
In this study, the team determined the three-dimensional structure of the complex formed by PTH1R and a G protein called Gq using cryo-electron microscopy. Structural analysis revealed that the intracellular loop 2 of PTH1R plays a crucial role in the selection of G proteins.
These findings not only advance our understanding of calcium homeostasis mechanisms in higher animals but also hold promise for the development of osteoporosis therapeutics with fewer side effects.
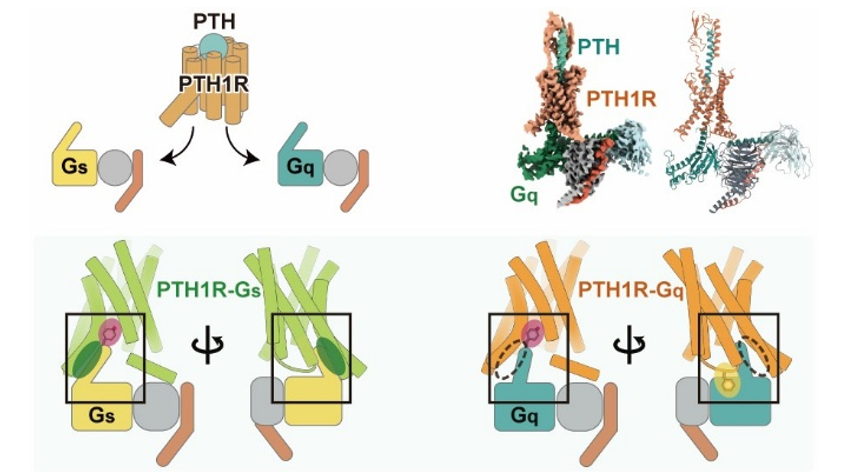 Figure: The structure of the channelrhodopsin KnChR obtained by cryo-electron microscopyG Protein Selectivity Mechanism of PTH1R Revealed by Its Three-Dimensional Structure
Figure: The structure of the channelrhodopsin KnChR obtained by cryo-electron microscopyG Protein Selectivity Mechanism of PTH1R Revealed by Its Three-Dimensional Structure
Journals
-
Journal name Nature Chemical BiologyTitle of paper Insights into G-protein coupling preference from cryo-EM structures of Gq-bound PTH1R


