Is it not a violation of the conservation of energy?
Imagine shining low-energy light with a long wavelength on a certain substance and seeing it “transform” into high-energy light with a short wavelength as it is reflected. If that happened, you might think it was a violation of the law of the conservation of energy. A certain amount of energy must be lost when light encounters a substance. Therefore, it should not be possible for light to be reflected with more energy than it had in its previous state.
However, the “certain substance” created by Nobuhiro Yanai and his team reflects light with higher energy than the light it received. How could such a phenomenon occur? How can one turn into two?
“It is not that one turns into one. Rather, one plus one equals two. Two similar molecules receive energy, and when they meet, their energies combine, emitting light. This phenomenon, which appears as if 1 turned into 2, is called photon upconversion. However, the result is not exactly one plus one equaling two. Some energy does get lost. Conceptually, it is closer to one plus one becoming one and a half.”
Yanai and his collaborators have already succeeded in converting infrared light into visible light and visible light into ultraviolet light. They are truly global leaders in the field of photon upconversion. Their technology is drawing significant attention as it holds promise for innovation in various fields such as solar power generation and artificial photosynthesis.
Let us delve a bit deeper into the mechanism of photon upconversion.
“When a molecule absorbs light, an electron becomes excited (enters a higher energy state) and moves to the next, higher orbital. Normally, electrons exist in pairs. As the electron moves to a higher orbital, the spins of the two electrons (a quantum property related to magnetism, with only two possible orientations: up or down) align in the opposite direction 《↑↓》 and cancel each other out. This is called the “excited singlet state.” When the electrons return to the original state (ground state), they release the excess energy as light. However, their light vanishes instantly, making it impossible to combine their energies.”
The researchers had to find a way to make the light emitted last longer if they were to stand a chance of combining them. To achieve this feat, they had to get the molecules into a “photoexcited triplet state.” What changes when the singlet state turns into a triplet state? The terms themselves sound complicated, but simply put, the triplet state is where the electron spins change from the singlet state's 《↑↓》 to all pointing in the same direction, like 《↑↑》 or 《↓↓》.
“Molecules in the photoexcited triplet state have had their spins flipped once, so it takes a long time for them to return to their original ground state, lasting over a thousand times longer than the singlet state. During this time, when triplet molecules encounter other triplet molecules, they exchange electrons. The light that they emit when they return to their original state has a higher energy than before because of the excess energy they accumulated incorporating electrons.”
This sounds easy in theory, but making it happen is where the rubber hits the road. Making molecules shine according to theory is an extremely difficult feat, and researchers worldwide had been scratching their heads after numerous trials and the same number of errors. It was Yanai and his team who finally achieved a breakthrough.

Creating substances that do not exist (yet)
“The mechanism itself was known before, but it was extremely inefficient and not practically useful. For example, it was unclear how to generate ultraviolet light from visible blue or green light for photoreactions requiring high energy, like antibacterial or antiviral applications. It was also unclear what kind of molecules should be designed to achieve an efficient upconversion. No one had the answer.”
In search of that answer, Yanai and his team designed, synthesized, and experimentally tested numerous molecules. They performed quantum chemical calculations (computations elucidating the behavior of electrons within molecules) on hundreds of molecules to predict what properties a given molecular structure would exhibit. Then, they synthesized these new molecules.
“How do we convert visible, blue light into ultraviolet light? The mechanism is extraordinarily complex, so the number of parameters we must consider when designing molecules is enormous. Simply put, we needed two types of molecules: a chromophore molecule that absorbs light and converts into a triplet state, and a chromophore molecule that combines with it and emits light. Both had to be designed appropriately. By designing these molecules with extreme precision, we achieved highly efficient conversion of visible light into ultraviolet light.”
Yanai says the process of creating a substance that did not exist was a continuous struggle.
“It was a cycle of creating, testing, and if it did not work, redesigning it all over again. Finding the right one on the first try is impossible, so it was an extremely time-consuming task. There were many failures, but each failure was a learning experience. The hardest part, though, was having to continue without knowing when we would reach the goal.”
What sustained them through this grueling journey was a shared conviction within the team.
“Doing something no one else in the world has done is fascinating and creates new value. I think it was thanks to this shared conviction that we found the resolve to keep going without giving up. Failure is inherent in research. But if everyone on the team holds onto that feeling, we can overcome failure no matter how many times it happens.”
Many challenges remain before photon upconversion can be applied to solar power generation and artificial photosynthesis. Issues such as efficiency and requiring strong light for proper functioning remain. However, Yanai and his team continue to work on resolving these issues.

What chemists can do in the quantum age
The story is not over yet. There was a reason for the thorough description of photoexcited triplets and how electron spins align at the beginning. However, after his extensive research in photochemistry, Yanai began contemplating how he could leverage light and the spins of electrons to pioneer new frontiers in nuclear hyperpolarization and quantum sensing. These endeavors have now become the main research themes of his laboratory.
“Some call the current developments the second quantum revolution. Thus, we need to ask ourselves: what can we achieve and how can we contribute as chemists who design and create matter in this quantum era? We can design substance structures with extreme precision at the atomic level and generate spins within them. Moreover, we can freely alter molecular structures and even embed complex programs within molecules. We aim to explore what is possible in this quantum era using such techniques and abilities. That is the direction we are heading in recently.”
First, nuclear hyperpolarization. Most of the readers are probably familiar with MRI. It is a large, cylindrical machine used in hospitals for detailed examinations of internal organs and bones. What an MRI observes is nuclear spin. Specifically, it observes the spin of hydrogen nuclei within water molecules in the human body. However, as explained with photoexcited singlet and triplet states, the spin orientation can point up or down. Within the human body, the nuclear spins of hydrogen atoms are almost equally divided between up and down. Consequently, since spin is a source of magnetic force, the up and down spins cancel each other out, making the magnetic force barely detectable. However, if even one out of tens of thousands of spin pairs remains uncancelled, in a state called “polarization,” then that one magnetic force can be observed. In other words, MRI detects and images only the information represented by the difference in the number of up-spin and down-spin particles. This means that what the MRI actually measures contains an incredibly small amount of information.
Hyperpolarization aims to increase sensitivity by reducing the number of oppositely aligned spins that cancel each other out.
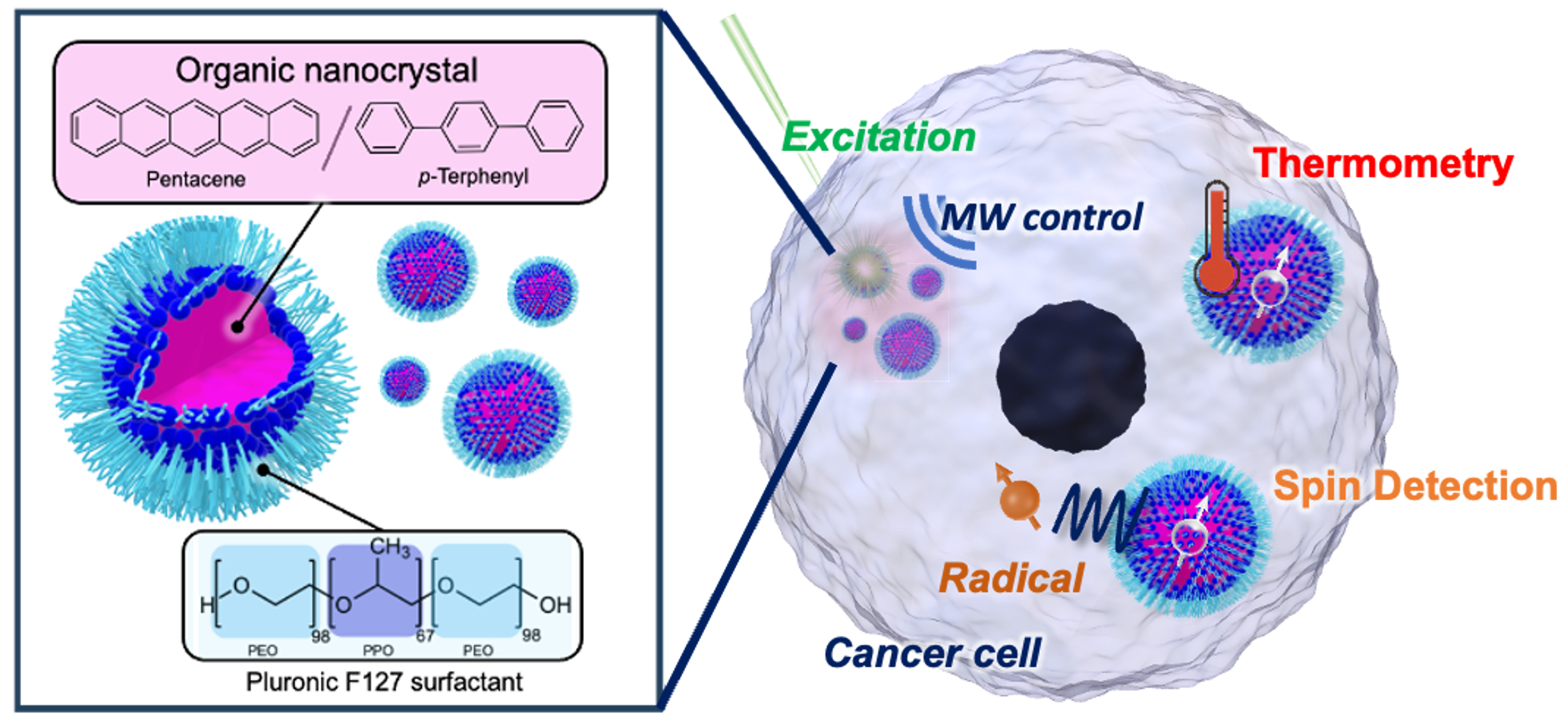
“This field has a long history. Currently, clinical trials are underway in the US and elsewhere. The method nearing practical application uses electron spins. It involves aligning the spins of electrons upward, then transferring this alignment to carbon nuclei, which are then injected into the patient’s body. However, this requires cooling to below -270 degrees Celsius to align the electron spins, making the equipment expensive and large-scale. The method we are developing can be used at room temperature and requires little energy from light. We have been working extensively on using photons and upconversion to “nudge” molecules into an excited state. Exploiting this excited state, we can generate spins and create polarization simply by shining light at room temperature. We hope to use this method to enhance MRI sensitivity.”
When asked how this would work in more practical terms, Yanai describes it as “shining light on something while you are microwaving it.”
“We enhance the sensitivity of the substance outside the body using electromagnetic waves and light, dissolve it in liquid, and inject it into the body. Before injecting it, we transfer the polarized electron spins to the nuclear spins. It is a hyperpolarized contrast agent-like substance.”
The frontier of quantum biochemistry
Furthermore, Yanai aims to make this hyperpolarized contrast agent-like substance easy to handle. His goal is to achieve high sensitivity at the target location for MRI imaging simply by spraying the substance onto, rather than injecting it into, the area of concern on the body and shining light on the contrast agent. In other words, he aims to make it as easy to use as examining an affected area with a gastrocamera.
“Surely, there are many occasions when doctors looking at MRI scans would want to see more detail in certain areas to diagnose the patient. However, this is currently impossible. If the substance we created could be used as a spray on the body over the area of interest inside the body, doctors would be able to shine light, literally and figuratively, on the state of the area of interest. That is what we hope to achieve using the power of chemistry to design molecules.”
By shining light on molecules to generate spins and prolonging their state to efficiently transfer them to target molecules, Yanai and his team have pursued this line of basic research for years. They can now achieve nuclear hyperpolarization with extremely high degree. Moreover, they discovered that the material developed to achieve this polarization can also be used for other quantum sensing applications.
“For example, a quantum sensing technique called optically detected magnetic resonance (ODMR) is used to measure the temperature inside living cells. Currently, this is done by inserting nanoscale diamonds into the cells. However, if we use the molecules we created as quantum sensors, the molecules, due to their uniformity, can provide a much more precise indication of the local temperature. Using molecules for quantum sensing is a very novel approach. I feel this could lead to the development of a large new field.”
Yanai says the originality of his research lies in his entry into the field of quantum physics as a chemist with the skillset to create new molecules and materials. His approach makes it possible to develop new quantum sensors that can be used for studying life phenomena and medical applications. He calls this emerging frontier quantum biochemistry.
“It expresses the goal of leveraging chemistry to bridge the gap between quantum and biological phenomena, two seemingly distant realms. Chemistry allows us to precisely define and design the quantum states of molecules down to each atom. Moreover, molecules are small and have low toxicity, making them suitable as probes within living organisms. In other words, I see chemists who can design and create molecules as the people who can help understand life phenomena by controlling quantum phenomena.”
Feel alone
As a Kyoto University graduate, Yanai was immersed in MOF research (materials with microscopic pores, where just 1 gram has a surface area equivalent to a soccer field) from his undergraduate days through his doctorate, mentored by none other than the Nobel laureate Professor Susumu Kitagawa.
“Professor Kitagawa often told me to “feel alone.” He meant that to mean not to overfocus on MOFs, but to step outside that world, to go where everything is unknown, where everyone is unfamiliar, and compete there. He taught me that this is how the originality of one's research is born. He instilled in me the values of never being a follower, always being a pioneer, and not to miss the forest for the trees.”
He says he wants to be able to look back and say that he spent his research life challenging himself to pioneer new fields.
So, what advice does he have for the next generation?
“I think it is perfectly fine if you have not yet found your life goals or dreams as a high school or undergraduate student. There is no need to rush. Keep challenging yourself with things you find interesting. If you continue to do so, somewhere along the way, your “this is it” moment will come. When that happens is different for everyone. It is best to face yourself without worrying too much about others. Finding something you want to dedicate your life to and being able to immerse yourself in it is surely a happy life. I hope you will take your time, without rushing, to find that thing.”
In his personal life, Yanai plays with his kids and goes jogging. He is also in charge of cooking on weekends.
“It seems such a cliche for a chemist to enjoy cooking. My specialty? My family often requests steamed buns. I make them from scratch, fermenting the dough and steaming them fresh. I think they are quite delicious,” he says proudly.
Coincidentally, if you squint a little, the Chinese characters for steam buns resemble the characters for molecules.
※Year of interview: 2026
Interview/Text: OTA Minoru
Photography: KAIZUKA Junichi

![リガクル[RIGAKU-RU] Exploring Science](/ja/rigakuru/images/top/title_RIGAKURU.png)










